|
Back to Blog
Tyrosine melanin dopamine drawit5/8/2023  
Blood amino acid quantification should be performed as a diagnostic follow-up of positive newborn screen. A metabolic physician should see newborns with elevated phenylalanine levels as soon as possible. A mean cutoff for Phe of 130 μmol/L (with a range of 65–234 μmol/L) and a Phe:Tyr ratio > 3 is abnormal. Elevated phenylalanine (Phe):tyrosine (Tyr) ratio over 2.5 mg/dL increases the likelihood of PKU. Elevated phenylalanine level can be caused not only by PKU but also defects of tetrahydrobiopterin, prematurity, liver disease and high protein intake. Newborns with Phe levels higher than 2 mg/dL (120 μmol/L) are reported as a positive screen. Many laboratories also calculate and report the phenylalanine and tyrosine ratio to increase the specificity of the screening test. Phenylalanine levels are measured with tandem mass spectrometry (MS/MS) using dried blood spots in all newborns in US and many developed countries as early as 24 hours of life. Today, PKU is screened in all states of the USA and many other countries in all over the world allowing early diagnosis and better health outcome as well as resulting in less economic burden on the health system. It was very successful, and many other states mandated the screening by the mid-1960s in the USA. 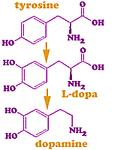
Newborn screening for PKU was first started in Massachusetts in 1963. The test was fast, simple and inexpensive, and had acceptable specificity and sensitivity. In 1962, Robert Guthrie developed a bacterial inhibition assay to measure phenylalanine level on a filter paper. About 20 years later, a German professor, Horst Bickel, developed the first Phenylalanine- free formula that could decrease blood phenylalanine levels and prevent intellectual disability. He then used chemical analysis to determine the nature of the substance that caused that deep green color change and extracted phenylpyruvic acid from their urine. He added ferric chloride to the urine of two children with intellectual disability and observed the deep green color change in their urine sample. Foelling, a Norwegian physician discovered PKU in 1934. Hyperphenylalaninemia, mild tremor, neonatal hypotonia, high neopterin, subnormal biopterin, Primapterin is presentĭr. Pterin-4-carbinolamine-dehydratase deficiency 
Microcephaly, hypotonia, mental retardation, seizures, hyperphenylalaninemia, normal/high neopterin, very high biopterin L-dopa/carbidopa,5-OH-tyrptophan, folinic acidĭihydropteridine reductase (DHPR) deficiency Mental retardation, microcephaly, seizures, irritability, abnormal movements, hyperthermia, hyperphenylalaninemia, high neopterin, trace biopterinĭopa-responsive dystonia, sepiapterin is present in urine, CSF Liver, brain, kidney, lymphocytes, erythrocytes, fibroblasts Low-phe diet, BH4, L-dopa/carbidopa,5-OH-tyrptophan, folinic acidĦ-Pyruvoyl-tetrahydropterin synthase deficiency Seizures, hypotonia, tremor, developmental delay, hyperphenylalaninemia, very low neopterin and biopterin In addition to kinetic feedback inhibition and the formation of an inhibitory dopamine/Fe+3 complex, these findings suggest that a third mechanism exists by which dopamine (or DOPA) can inhibit tyrosine hydroxylase, adding further complexity to the regulation of catecholamine biosynthesis.Inherited disorder of phenylalanine metabolism Enzyme defects Furthermore, the covalent modification and inactivation of tyrosine hydroxylase was blocked by antioxidant compounds (dithiothreitol, reduced glutathione, or NADH). Inactivation was associated with covalent incorporation of radiolabelled dopamine into the enzyme as assessed by immunoprecipitation, size exclusion chromatography, and denaturing sodium dodecylsulfate (SDS)-polyacrylamide gel electrophoresis. Catecholamine-mediated inactivation occurred with both purified tyrosine hydroxylase as well as enzyme present in crude pheochromocytoma homogenates. Dihydroxyphenylalanine (DOPA the catechol-containing precursor of dopamine) also inactivated tyrosine hydroxylase under these conditions. The present studies demonstrated that dopamine quinone, the formation of which was enhanced through the activity of the melanin biosynthetic enzyme, tyrosinase, covalently modified and inactivated tyrosine hydroxylase. One of the potential targets in brain for dopamine quinone damage is tyrosine hydroxylase, the rate-limiting enzyme in catecholamine biosynthesis. This oxidation can be enhanced through various enzymes including tyrosinase and/or prostaglandin H synthase. Dopamine can be oxidized to form a reactive dopamine quinone that can covalently modify cellular macromolecules including protein and DNA. Dopamine has been implicated as a potential mediating factor in a variety of neurodegenerative disorders. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
